|
1/18/2024 0 Comments Fission reaction equation examples
Acting in opposition is coulombic repulsion term, which acts to increase the distance between repelling proton pairs and thus promotes elongation of the nucleus into an oval shape. /GettyImages-103311355-490ad3ba66d44d40b738a0e7d468ac8a.jpg) 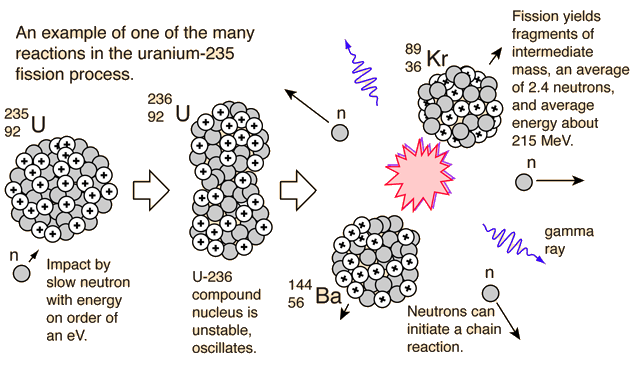
In this model, as with a classical liquid drop, a " surface tension" term is introduced which promotes the spherical shape of the nucleus. : 35 The semi-classical liquid drop model provides a primarily qualitative description of the phenomenology by treating the nucleus as a classical drop of liquid to which quantum corrections can be applied, which provides a useful conceptual picture that matches in part with experimental data, but ignores much of the quantum nature of the system and fails at more rigorous predictions. : 3 Computational theories based on Hartree-Fock or density-functional theory approaches have been developed, however computational complexity makes it difficult to reproduce the full behaviour. To date, true ab initio models describing the complete fission process are not possible. : 4 The barrier to fission is expected to be zero at A=300, though an island of stability may exist centred around Z=114, N=184. Shell effects may further affect observed half-lives - decays of odd-A nuclides are hindered by 3-5 orders of magnitude compared to even-even nuclides. As nuclear mass increases, so too does the fissility parameter, eventually approaching and exceeding unity, where stability against fission is lost altogether. : 3 For light nuclei, x is small and a sizeable fission barrier exists. The stability of a nuclide against fission is expressed as the ratio of the Coulomb energy to the surface energy, which can be empirically estimated as the fissility parameter, x: Such a barrier is energetically possible for all A>100, though its height generally decreases with increasing Z, : 433 and fission is only practically observed for A>232. Instead it must tunnel through a potential barrier, with a probability determined by the height of the barrier. Spontaneous fission is usually a slow process, as the nucleus cannot simply jump to the lower energy (divided) state. Thus, at high mass and proton numbers, coulombic repulsion overpowers the nuclear binding forces, and the nucleus is energetically more stable as two separate fragments than as a single bound system. 
Nuclear binding energy increases in proportion to atomic mass number (A), however coulombic repulsion increases with proton number (Z) squared. Spontaneous fission arises as a result of competition between the attractive properties of the strong nuclear force and the mutual coulombic repulsion of the constituent protons. Such an effect could only be explained by spontaneous fission of the uranium nuclei without external influence. The discovery of induced fission itself had come as a surprise, and no other mechanism was known that could account for the observed decays. Their equipment recorded fission fragments even when no neutrons were present to induce the decay, and the effect persisted even after the equipment was moved 60m underground into the tunnels of the Moscow Metro's Dinamo station in an effort to insulate it from the effects of cosmic rays. Following the discovery of induced fission by Otto Hahn and Fritz Strassmann in 1938, Soviet physicists Georgy Flyorov and Konstantin Petrzhak began conducting experiments to explore the effects of incident neutron energy on uranium nuclei.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
